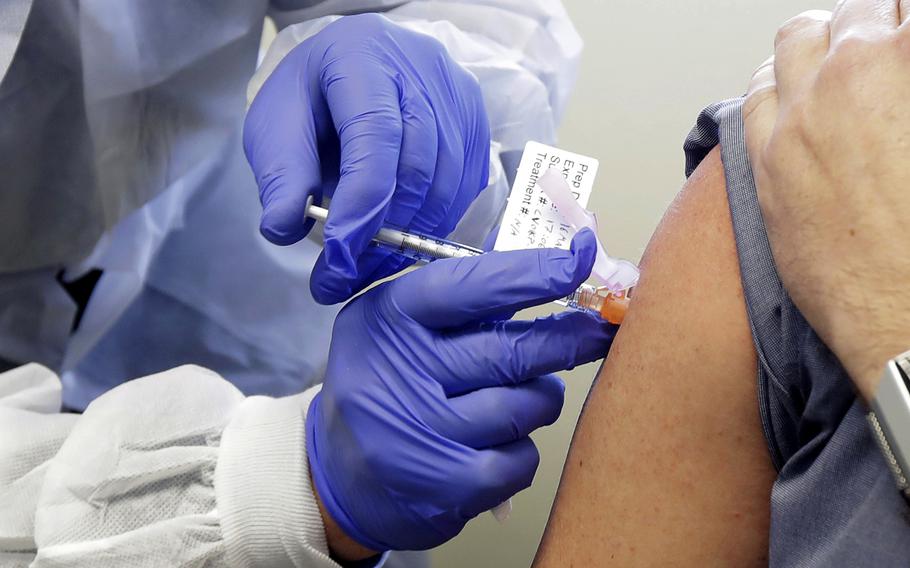
In this March 16, 2020 file photo, a subject receives a shot in the first-stage safety study clinical trial of a potential vaccine by Moderna for COVID-19, the disease caused by the new coronavirus, at the Kaiser Permanente Washington Health Research Institute in Seattle. (Ted S. Warren/AP)
Stars and Stripes is making stories on the coronavirus pandemic available free of charge. See other free reports here. Sign up for our daily coronavirus newsletter here. Please support our journalism with a subscription.
U.S. service members will have to volunteer if they want early access to coronavirus vaccines under emergency or early use guidelines set by the Food and Drug Administration, according to the Defense Department.
At least three vaccines are better than 90% effective against COVID-19, the respiratory disease caused by the coronavirus, according to news accounts. The U.S. military is planning to distribute two vaccines that are pending FDA approval.
Vaccines could become available soon. The FDA takes up a request Thursday for emergency use of the vaccine developed by Pfizer-BioNTech, according to the FDA docket online. The United Kingdom last week approved its use on a temporary, emergency basis, the first country to do so.
Moderna has also applied for permission to field its vaccine on an emergency use basis. An FDA committee meets on that application Dec. 17.
Vaccines available on a trial basis will probably be administered first to volunteers, according to a Defense Department statement given to Stars and Stripes on Thursday by Yokota Air Base in western Tokyo.
The Defense Department expects quantities will be “limited initially and distributed on a rolling delivery basis as more vaccines become available,” according to the email from base spokesman Lt. Stuart Thrift.
Inoculations will be voluntary until the vaccines are finally approved by the FDA, according to the DOD statement he provided.
Anyone who requests the vaccine will be required to give informed consent, meaning they must confer with their doctor to receive it, according to a Nov. 26 report by Military.com. The report quoted Col. Jessica Spitler, 30th Medical Group commander, who spoke at a town hall on Nov. 6.
Inoculations may remain voluntary as long as two years, Military.com quoted Spitler as saying.
On Wednesday, Chief of Naval Operations Adm. Mike Gilday said military health providers and first responders will be at the head of the line when vaccines become available for the military.
Gilday, speaking before a Senate Armed Services Committee subpanel, said the Defense Department’s vaccines are being sent to every military medical treatment facility within the United States as well as locations overseas.
Locations in the U.S. will get the Pfizer vaccine, which has to be kept at -70 degrees Celsius and is good for five days after being thawed, he said. Overseas installations will get the Moderna vaccine, which is good for 30 days with standard refrigeration at 2 to 8 degrees Celsius.
Health care professionals and then emergency and safety personnel at installations will have priority, which could include DOD civilians and contractors, according to the Centers for Disease Control and Prevention.
ditzler.joseph@stripes.com Twitter: @JosephDitzler